The therapeutic landscape of MASLD is undergoing a fundamental shift. With the recent approval of resmetirom (Rezdiffra™) and the continued development of agents such as GLP-1 receptor agonists, MASLD is moving from a condition managed primarily through lifestyle intervention to one increasingly treated with targeted pharmacological therapies.
This transition introduces a critical question:
“How can treatment response be assessed in a disease where clinical symptoms are limited and traditional biomarkers are poorly aligned with underlying pathology?”
Mechanistic Complexity of Treatment Response in MASLD
MASLD is not a single-pathway disease. It is characterized by a complex interplay of:
- – Hepatic lipid accumulation (steatosis)
- – Insulin resistance and metabolic dysregulation
- – Inflammatory signaling
- – Hepatocyte injury and apoptosis
- – Fibrogenesis and extracellular matrix remodeling
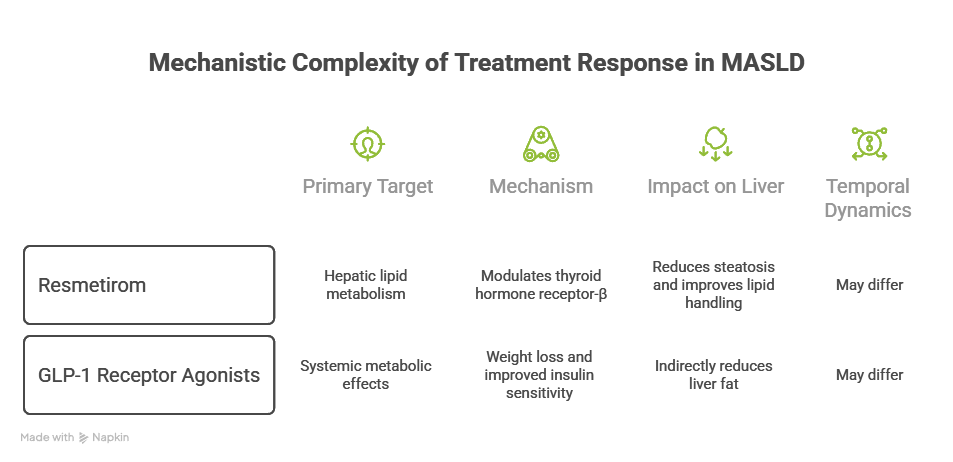
Pharmacological therapies target different aspects of this spectrum. Resmetirom (a thyroid hormone receptor-β agonist) primarily modulates hepatic lipid metabolism, reducing steatosis and improving lipid handling. In contrast, GLP-1 receptor agonists such as semaglutide exert systemic metabolic effects, including weight loss and improved insulin sensitivity, leading indirectly to reductions in liver fat.
While these therapies may ultimately converge on improving liver histology, their mechanisms of action differ, and so too may the temporal dynamics of response.
Limitations of Conventional Monitoring Endpoints
Biochemical Markers
Serum transaminases (ALT, AST) are widely used in clinical practice but have well-documented limitations. They lack sensitivity for detecting ongoing hepatocyte injury, and changes in ALT may not correlate with histological improvement, particularly in patients with advanced fibrosis. In addition, normal ALT levels are frequently observed in patients with active MASH.
Imaging-Based Assessments
Imaging modalities such as MRI-PDFF and elastography provide valuable information on hepatic fat content and liver stiffness as a proxy for fibrosis. However, reductions in liver fat do not necessarily reflect resolution of inflammation or hepatocyte injury. Furthermore, fibrosis regression is typically slow, limiting the utility of these tools as early treatment endpoints.
Histological Endpoints
Liver biopsy remains the reference standard in clinical trials, enabling assessment of steatohepatitis resolution and fibrosis stage. However, biopsy is constrained by sampling variability and Invasiveness and is hence impractical for real world serial monitoring
A Gap Between Treatment and Decision-Making
While pharmacological treatment options are expanding, clinical frameworks for monitoring and decision-making remain under development. In routine practice, questions such as:
- – When should a therapy be considered ineffective?
- – When should treatment be escalated or switched?
- – What constitutes a meaningful response?
are not yet clearly defined in current guidelines.
This creates a disconnect between the availability of therapies and the ability to act on treatment response, highlighting the need for monitoring approaches that can provide more informative and dynamic insights into disease activity.
Biomarkers in Clinical Trials: Capturing Dynamic Disease Activity
Given these limitations, there has been increasing interest in circulating biomarkers that reflect active disease processes, particularly in early-phase treatment response.
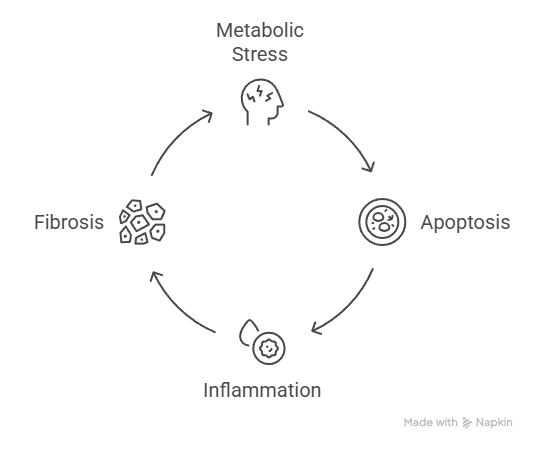
One of the key pathological features of MASLD progression, especially in MASH, is hepatocyte apoptosis. This process contributes directly to inflammatory signalling, activation of stellate cells, and fibrogenesis.
Fragments of caspase-cleaved keratin-18, released during apoptosis, can be detected in circulation and quantified using assays such as M30 Apoptosense® ELISA.
M30® in Pharmacological Studies
M30® has been incorporated into multiple MASLD clinical studies as a non-invasive biomarker of hepatocyte injury and treatment response.
- – Reductions in M30® levels have been associated with improvements in histological features of disease activity, including ballooning and inflammation (Davison et al., Journal of Hepatology, 2021).
- – In interventional settings, M30® has demonstrated responsiveness to therapeutic modulation, reflecting reductions in apoptotic activity in parallel with metabolic and histological improvements.
Importantly, M30® captures a dimension of disease activity that is not directly assessed by imaging or standard biochemical tests; the rate of ongoing hepatocyte injury.
Temporal Aspects of Treatment Response
One of the key advantages of biomarkers reflecting apoptosis is their ability to detect early biological changes.
Pharmacological interventions may rapidly influence lipid metabolism or insulin sensitivity, but the downstream effects on inflammation and fibrosis may take longer to manifest. Apoptosis sits at an intermediate point in this cascade, downstream of metabolic stress but upstream of fibrosis, making it a potentially sensitive marker of early response.
This positions biomarkers such as M30® as useful for:
- – Early signal detection in clinical trials
- – Assessing biological response before structural changes become evident
Toward Integrated Monitoring Strategies
No single marker fully captures the complexity of MASLD. Instead, a multi-modal approach is likely required, combining structural assessment (imaging and fibrosis scores), functional markers (liver enzymes), and mechanistic biomarkers such as apoptosis markers.
Within this framework, M30® can provide insight into ongoing hepatocyte injury, complementing other tools that assess steatosis or fibrosis.
Looking Ahead
As pharmacological therapies become more widely used in MASLD, the need for sensitive, non-invasive, and mechanistically relevant biomarkers will continue to grow.
Beyond detection, future monitoring strategies may also need to support clinical decision-making, helping to identify non-responders and guide therapeutic adjustments, areas where current guidance remains limited.
In the next post in this series, we will explore in more detail the biological role of hepatocyte apoptosis in MASLD, and why it represents a key target for both disease understanding and monitoring.