News
17 April 2024
Home Slider
K18 – the Key to Unlocking Informed Decisions in MASH Drug Trials
Join us for an enlightening webinar presented by Jessica Tuohy, a distinguished partner and colleague from the US, focusing on the significance of K18 as a biomarker in the development of treatments for MASH (Metabolic Associated Steatohepatitis). This webinar is essential for drug developers, doctors, researchers, and key opinion leaders in the field of metabolic liver diseases, offering deep insights and key research findings.
Event Details:
- Date: Tuesday, April 23
- Time: [1pm EDT, 7pm CET]
Webinar Overview: This session will cover several critical areas:
- – An Introduction to the Growing Epidemic of MASLD (Metabolic Associated Steatotic Liver Disease)
- – Challenges in Developing Therapies for the Treatment of MASH
- – An Overview of K18 and Its Role in MASH Pathogenesis
- – A Review of Data That Support the Value of K18 as a Biomarker for Assessing the Efficacy of Drug Candidates in MASH Clinical Trials
Who Should Attend:
- – Drug developers in the field of MASLD and MASH.
- – Physicians and healthcare professionals specializing in hepatology, gastroenterology, and related fields.
- – Medical researchers focusing on liver disease and drug development.
- – Key opinion leaders in metabolic liver diseases.
Registration: Please register here to secure your spot in this informative webinar. Registration is required to access this online event.
Additional Information: If you are unable to attend the live event, a recording will be made available to all registered participants, ensuring you can access this webinar at your convenience.
We look forward to welcoming you to what promises to be an informative and transformative session.
For more information, please contact us at marketing@vlvbio.com.
Don’t miss out on the chance to advance your knowledge in MASH treatment and diagnostics. Reserve your spot today!
23 January 2024
Blog
Launch of IMMUNIS Cytokeratin 18F EIA by Institute of Immunology, Japan
We are thrilled to share a momentous achievement in our longstanding collaboration with our esteemed Japanese partners, the Institute of Immunology. This announcement comes during a meeting filled with collaboration and shared successes as Mr. Ito, Mr. Akira, and Ms. Yukiko from the Institute of Immunology paid a visit to our headquarters in Stockholm.
During this collaborative visit, we had the pleasure of discussing plans for the imminent launch of the IMMUNIS Cytokeratin 18F EIA, a project that holds significant promise in the realm of diagnostic medicine. This project has been a work of dedication and innovation, and the collaboration between VLVbio and the Institute of Immunology has been instrumental in its success!
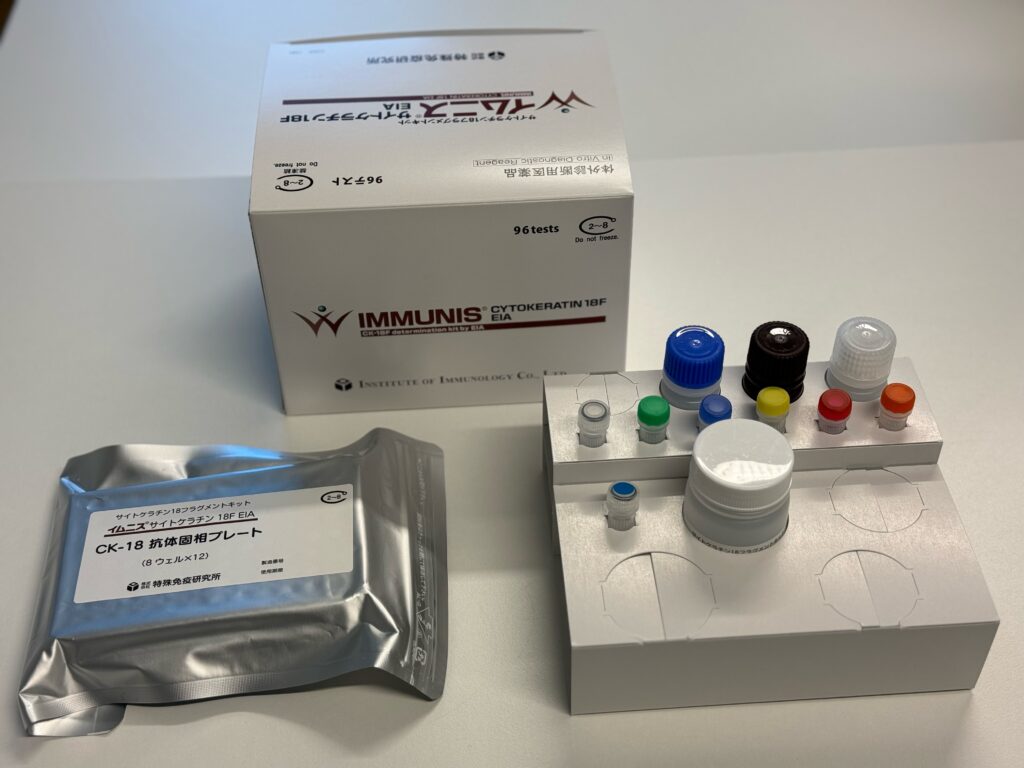
We are happy that the IMMUNIS Cytokeratin 18F EIA has gained official approval as an In Vitro Diagnostic (IVD) for the diagnosis of Non-Alcoholic Steatohepatitis (NASH) from the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan! This recognition underscores the product’s efficacy and reliability in contributing to the diagnosis and management of NASH, a condition of growing concern globaly as the epidemiology of the disease keeps increasing at an alarming rate. The need for a biomarker to identify patients with NASH and who are at risk of significant liver disease is of utmost importance, and we are happy that the Keratin 18 assay can assist in that!
The IMMUNIS Cytokeratin 18F EIA ELISA kit
Of equal importance, the IMMUNIS Cytokeratin 18F EIA, developed by the Institute of Immunology, has been included in the national health insurance system in Japan. This recognition ensures widespread accessibility, facilitating broader usage of this diagnostic tool within the Japanese healthcare community.
We are pleased to announce that the official launch date for the IMMUNIS Cytokeratin 18F EIA is scheduled for February 26th to the Japanese market. This date signifies the conclusion of a meticulous research and development process and exemplifies the collaborative efforts between VLVbio and our esteemed partners at the Institute of Immunology. The IMMUNIS Cytokeratin 18F EIA, launched by the Institute of Immunology, is poised to make a substantial impact on the field of NASH diagnosis, providing healthcare professionals with an additional tool to enhance their diagnostic capabilities in Japan.
As we celebrate this milestone and the successful visit from our partners, we extend our gratitude to all individuals who have contributed to the realization of this project, including the dedicated team members and esteemed partners at the Institute of Immunology. Congratulations to you all!

Institute of Immunology president Mr. Ito Yukio and VLVbio CEO
Mrs. Slavica Brnjic holding the finished IMMUNIS Cytokeratin 18F assay.
9 April 2026
Blog
Monitoring MASLD: Why Assessing Treatment Response Remains a Clinical Challenge
Metabolic Associated Steatotic Liver Disease (MASLD) is increasingly recognized as a major global health concern, closely linked to metabolic conditions such as obesity and Type 2 diabetes. While awareness and diagnostic capabilities have improved in recent years, managing MASLD remains challenging, particularly when it comes to assessing treatment response over time.
With the therapeutic landscape evolving, including the introduction of pharmacological treatments alongside established lifestyle interventions, clinicians are now faced with an important question: how can we reliably monitor whether a patient is improving?
The Challenge of a Silent and Dynamic Disease
MASLD often progresses without clear symptoms, especially in its early and intermediate stages. Patients may feel well even as underlying liver injury continues. This disconnect makes objective monitoring tools essential, particularly when evaluating the effectiveness of treatment.
At the same time, MASLD is not a static condition. Disease activity can fluctuate depending on metabolic control, weight changes, or therapeutic interventions. As a result, clinicians require tools that can capture ongoing biological processes, rather than relying solely on static or structural assessments.
Limitations of Current Monitoring Approaches
Liver Enzymes (ALT, AST)
Liver enzymes are widely used in clinical practice but have important limitations. Many patients with MASLD may present with normal ALT and AST levels despite ongoing liver injury, while others may show fluctuations that do not reflect meaningful histological changes. As such, liver enzymes alone are often insufficient to monitor treatment response.
Imaging Techniques
Non-invasive imaging tools, such as ultrasound or elastography, provide valuable information about liver fat and fibrosis. However, these methods primarily assess structural changes and may not capture early or dynamic changes in disease activity, particularly in response to short-term interventions.
Liver Biopsy
Liver biopsy remains the reference standard for assessing disease activity and fibrosis. However, its use in routine follow-up is limited due to:
- – Invasiveness and patient burden
- – Sampling variability
- – Cost and logistical constraints
For these reasons, repeated biopsies are not practical for monitoring treatment response in most patients.
A Growing Need in the Era of MASLD Treatment
The need for reliable monitoring tools has become even more pressing as MASLD enters a new phase of active management. With the development of pharmacological therapies, alongside lifestyle and surgical interventions, clinicians must now evaluate:
- – Whether a treatment is effective
- – How early a response can be detected
- – Which patients may require adjustments in their management plan
This shift highlights a clear gap in current clinical practice: the lack of accessible, non-invasive tools that reflect ongoing liver injury and response to therapy.
Moving Toward Biological Markers of Disease Activity
To address these challenges, attention will increasingly turn toward biomarkers that reflect underlying disease mechanisms, rather than only structural liver changes.
One such mechanism is hepatocyte apoptosis, a key feature of MASLD progression, particularly in metabolic dysfunction-associated steatohepatitis (MASH). Measuring this process may provide a more direct insight into active liver injury and how it changes over time.
Biomarkers targeting apoptosis, such as those detecting fragments of keratin-18, have therefore been explored as tools to support treatment monitoring and improve clinical decision-making.
Looking Ahead
As MASLD management continues to evolve, the ability to monitor treatment response accurately and non-invasively will be critical. Understanding how different tools reflect disease activity, and how they can be integrated into clinical practice, will play a central role in improving patient outcomes.
In the next post in this series, we will explore how treatment monitoring is becoming increasingly important in the context of emerging pharmacological therapies, and how biomarkers are being used in clinical trials to assess response.
11 December 2025
Home Slider
Thank you for being part of our 2025
Dear colleagues, partners, and friends,
As we conclude another remarkable year, we want to extend our sincere gratitude to each of you; our distributors, collaborators, and partners around the world.
2024–2025 has been a year defined by progress, dialogue, and renewed momentum in the MASLD/MASH field. We are truly grateful for the continued trust, engaged discussions, new scientific contacts, and the many constructive conversations we’ve shared with you throughout the year.
The global MASLD landscape is evolving faster than ever. The release of Rezdiffra™ in the United States has already brought meaningful benefit to many patients, marking a long-awaited step forward in treatment availability. With the recent approval in Europe, we are excited to see this progress finally reach European patients, which is a milestone the field has been anticipating for decades.
Likewise, the approval of semaglutide for MASH adds an important therapeutic option. After years of limited tools, we are now entering a new phase where patients with MASH can finally receive the help they need.
But as treatment availability expands, a new challenge has emerged:
How do we effectively monitor patients on therapy?
This question has been raised repeatedly in our conversations with clinicians, KOLs, and partners this year, and it is becoming one of the central needs in clinical practice.
In this evolving landscape, M30 Apoptosense® ELISA has shown clear potential to fill an important part of this gap.
Evidence from multiple clinical trials, including Madrigal’s phase III MAESTRO-NASH trial, has shown that M30® (ccK18) is one of the leading blood-based indicators of treatment response, outperforming several structural NITs in early prediction of change. Importantly, M30® offers:
- – Early, biology-based insight into hepatocellular injury
- – Reliable tracking of treatment effect before structural changes become visible
- – A practical solution for clinics without access to advanced imaging such as MRI-PDFF
- – A low-cost, accessible assay that integrates easily into routine follow-up workflows
As more patients in the US and Europe begin therapy, the need for simple, repeatable, blood-based monitoring tools has never been clearer. We are proud that our M30 Apoptosense® ELISA continues to serve as a scientifically grounded, widely studied option to support clinicians and improve real-world treatment follow-up.
Looking ahead to 2026, we are excited to continue supporting you; through scientific collaboration, distributor partnerships, educational materials, and tools that strengthen your local markets. Your work is essential for bringing knowledge, clarity, and better care to patients worldwide, and we value the role you play in this shared mission.
From all of us at VLVbio:
Thank you for a productive, inspiring, and forward-moving year.
We look forward to continuing our work in 2026.
The VLVbio Team

12 June 2025
Home Slider
Global Fatty Liver Day
1 in 3 adults has fatty liver disease. And many don’t even know it.
Today on Global Fatty Liver Day, we stand with the global health community to raise awareness of this silent epidemic and the power of early detection.
At VLVbio, we’re committed to supporting healthcare professionals with diagnostic tools that enable timely action. Read more on how the VLVbio products assist diagnosing steatotic liver diseases here!
Know it. Test it. Treat it. 💚
22 May 2025
Home Slider
Play For Your Liver
Liver disease isn’t just an adult problem.
Today, 1 in 10 children are at risk of MASLD (Metabolic dysfunction-associated steatotic liver disease), and the numbers are rising fast.
At VLVbio, we believe that prevention starts young – through movement, healthy habits, and community support.
That’s why we’re proud to sponsor a local Swedish youth football team, promoting an active lifestyle and early liver health awareness.
Because every goal scored is a step toward a healthier future! ⚽
Watch our short campaign video below to see how we’re helping raise awareness and protect the next generation, one active child at a time.
#PlayForYourLiver
25 April 2025
Home Slider
The EASL Congress 2025
We are heading to Amsterdam for the EASL Congress together with the European Association for the Study of the Liver!
We’re happy to yet again be exhibiting at #EASLcongress with EASL | The Home of Hepatology! We are happy to yet again be an industry partner for the biggest liver event in Europe!
The latest in hepatology is happening 📅 7–9 May.
🤝 Join us at the highlight of the year! Register today, and lets meet and discuss liver biomarkers at our booth.
19 March 2025
Blog
High-Risk Populations and Targeted Interventions in MASLD
Metabolic Associated Steatotic Liver Disease (MASLD) affects millions worldwide, but certain populations face significantly higher risks due to underlying metabolic conditions and lifestyle factors. Early identification and tailored interventions for these high-risk groups are critical in mitigating disease progression and improving outcomes.
In this post, we focus on identifying these populations and the role of personalized diagnostics and care strategies.
Who Is at Risk?
MASLD is closely linked to metabolic dysfunction, making individuals with certain conditions particularly vulnerable to the disease. Recognizing these high-risk groups allows healthcare providers to focus resources and interventions where they are needed most.
1. Individuals with Type 2 Diabetes
Research indicates that up to 70% of individuals with Type 2 diabetes also have MASLD, often progressing silently toward fibrosis or cirrhosis. The interplay between insulin resistance and liver fat accumulation accelerates liver damage in this group.
2. Patients with Obesity
Obesity is one of the strongest predictors of MASLD, affecting up to 90% of this population. Excess visceral fat contributes to liver fat deposition and inflammation, increasing the likelihood of progression to more severe disease stages.
3. Additional High-Risk Groups
· Individuals with Dyslipidemia: Elevated triglycerides and low HDL cholesterol are common in MASLD patients.
· Patients with Hypertension: Often part of metabolic syndrome, hypertension contributes to the overall disease burden.
· Family History of Liver Disease: Genetic predisposition can heighten MASLD risk.
Identifying these populations during routine care, such as diabetes or obesity management, provides an opportunity to diagnose MASLD early, even before symptoms appear.
The Importance of Tailored Diagnostics
For high-risk populations, non-invasive diagnostic tools are invaluable in stratifying patients by their risk of progression and guiding care decisions.
M30 Apoptosense® ELISA
Biomarkers like M30 Apoptosense® ELISA measure hepatocyte apoptosis, offering a non-invasive way to detect early liver injury. In high-risk groups, M30 can be used to:
· Identify patients likely to progress to advanced disease stages.
· Monitor the effectiveness of lifestyle or medical interventions.
· Provide a measurable outcome to guide therapeutic decisions.
The Benefits of Tailored Diagnostics
· Risk Stratification: Directs resources toward patients at highest risk, avoiding unnecessary testing for low-risk individuals.
· Proactive Monitoring: Allows clinicians to track changes over time and intervene when needed.
· Cost-Effectiveness: Reduces healthcare expenditures by preventing advanced-stage complications.
By focusing on non-invasive, scalable tools, healthcare providers can effectively manage MASLD in high-risk populations without relying on resource-intensive methods like biopsies.
Targeted Interventions for High-Risk Groups
Once high-risk patients are identified, personalized interventions can significantly improve outcomes and prevent progression to severe disease stages.
1. Lifestyle Modifications
Lifestyle changes remain the cornerstone of MASLD management. Evidence shows that:
· Losing 7–10% of body weight can reduce liver fat, inflammation, and fibrosis.
· Adopting a Mediterranean diet can improve liver health by emphasizing whole foods, healthy fats, and reduced sugar intake.
· Structured exercise programs improve metabolic function and reduce liver fat.
2. Pharmacological Treatments
Emerging therapies targeting specific mechanisms of MASLD, such as liver fat accumulation or fibrosis, are becoming available. These treatments complement lifestyle changes in patients who require additional intervention.
3. Regular Monitoring
Tools like M30 Apoptosense® ELISA enable healthcare providers to track the impact of interventions and make timely adjustments. Regular follow-ups also reassure patients of their progress, encouraging long-term adherence.
Preventing the Burden of Advanced Liver Disease
High-risk populations are at an increased likelihood of progressing to cirrhosis, liver failure, or hepatocellular carcinoma. The long-term economic and clinical burden of these outcomes is substantial. By focusing on early diagnosis and targeted interventions:
· The progression to severe disease can be significantly reduced.
· Patient quality of life improves.
· Healthcare systems can avoid the high costs associated with advanced-stage treatments.
Conclusion
Targeting high-risk populations with tailored diagnostics and interventions is a critical step in addressing the global burden of MASLD. Non-invasive tools like M30 Apoptosense® ELISA, combined with lifestyle changes and emerging therapies, provide healthcare providers with the resources needed to deliver proactive, patient-centered care.
As MASLD continues to rise worldwide, adopting this targeted approach will play a pivotal role in improving outcomes and reducing the disease’s impact on patients and healthcare systems. Read more here!
